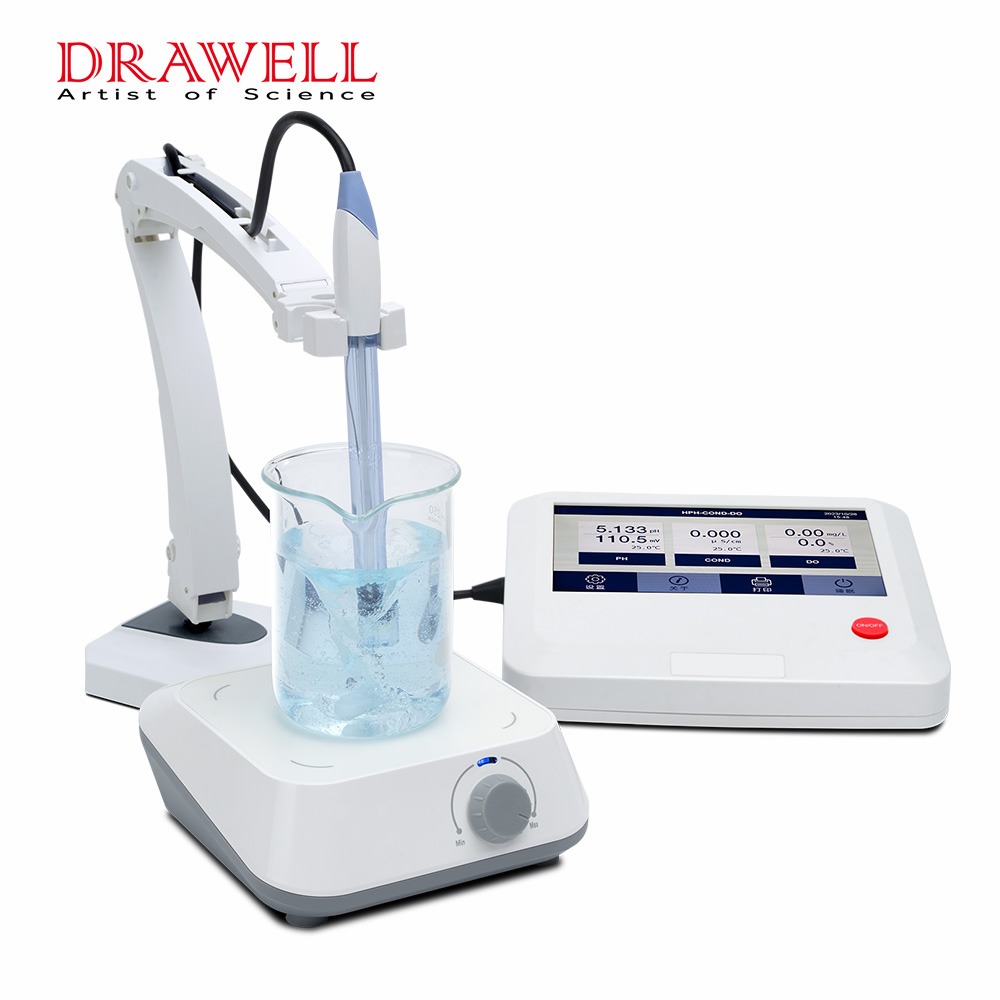
Conductivity meters are essential instruments used to measure the ability of a liquid to conduct electricity. These devices are widely used in various industries, including water quality monitoring, chemical processing, and laboratory research. Understanding how conductivity meters work can help users apply them effectively in different applications.
Working Principle of Conductivity Meters
Conductivity meters function based on the principle that an electrically conductive solution contains charged particles (ions) that enable the flow of electric current. The more ions present in the solution, the higher its conductivity. The meter applies an alternating electrical voltage to the solution and measures the resulting current flow, which is then converted into a conductivity value.
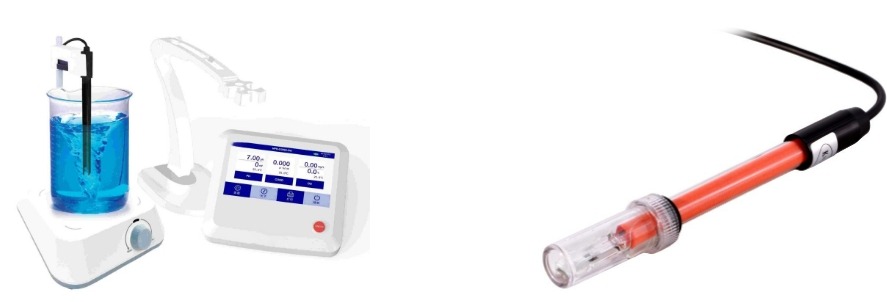
Key Components of a Conductivity Meter
A conductivity meter consists of several essential components that work together to measure the ability of a liquid to conduct electricity. Each component plays a critical role in ensuring accurate and reliable conductivity readings.
1. Electrodes (Probes)
Electrodes, also known as probes or sensors, are the primary components responsible for detecting conductivity. They are typically made of materials such as platinum, graphite, or stainless steel to resist corrosion and contamination. The electrodes are placed in the liquid sample, where they facilitate the flow of electrical current and measure the solution’s ionic concentration.
2. Voltage Source
A conductivity meter requires a voltage source to apply an alternating current (AC) to the electrodes. The AC voltage prevents polarization effects that could interfere with accurate readings. The strength of the applied voltage varies depending on the expected conductivity range of the solution.
3. Measuring Circuit
The measuring circuit detects the electrical current that flows through the solution in response to the applied voltage. This current is directly proportional to the number of dissolved ions in the liquid. The circuit then converts the detected current into a raw signal, which is further processed to determine the conductivity value.

4. Temperature Sensor
Since conductivity is temperature-dependent, most modern conductivity meters include a built-in temperature sensor. This sensor detects the sample’s temperature and applies automatic temperature compensation (ATC) to correct the conductivity reading, ensuring accuracy across different conditions.
5. Microprocessor and Display Unit
The microprocessor processes the electrical data collected by the measuring circuit and converts it into a readable conductivity value, typically displayed in microsiemens per centimeter (µS/cm) or millisiemens per centimeter (mS/cm). The display unit, often an LCD or digital screen, presents the final conductivity measurement in real-time. Advanced meters may also feature data logging, multi-parameter analysis, and connectivity options for transferring results to external devices.
6. Calibration System
Conductivity meters require regular calibration using standard conductivity solutions to maintain accuracy. Many meters have an automatic or manual calibration system that allows users to adjust the readings according to known reference values. Proper calibration ensures that the meter provides precise and reliable results over time.
Types of Conductivity Meters
Conductivity meters come in different types, each designed for specific applications and measurement requirements. The choice of a conductivity meter depends on factors such as the accuracy needed, the nature of the solution, and environmental conditions. This chart provides an overview of various types of conductivity meters, how they function, and the diverse applications they serve in different industries.
| Type | How Them Works | Applications |
| Contact (Electrode-Based) Conductivity Meters | Measures conductivity by directly applying an electrical voltage between two electrodes submerged in the solution. The ions in the solution conduct the current, which is then measured. | Water quality testing, laboratory analysis, industrial applications, environmental monitoring. |
| Inductive (Toroidal) Conductivity Meters | Uses an electromagnetic field to induce a current in the solution without direct contact. This prevents electrode fouling and corrosion. | High-conductivity solutions, chemical processing, wastewater treatment, corrosive environments. |
| Four-Electrode and Six-Electrode Conductivity Meters | Utilizes multiple electrodes to minimize polarization and improve accuracy, especially in highly conductive solutions. The measurement is more precise than standard two-electrode systems. | Scientific research, industrial solutions with extreme conductivity ranges, precise laboratory measurements. |
| Benchtop Conductivity Meters | Stationary, high-precision meters typically found in laboratories. They are used for precise measurements and often have additional features like temperature compensation. | Laboratory research, pharmaceutical testing, food and beverage quality control, chemical analysis. |
| Portable Conductivity Meters | Handheld devices designed for field measurements. They are battery-powered and offer ease of use for on-site testing. | Environmental monitoring, agriculture, on-site industrial testing, field analysis. |
| Online Conductivity Meters | Installed permanently in pipelines or tanks for continuous monitoring. They provide real-time data on conductivity levels. | Water treatment plants, chemical production, industrial process automation, continuous monitoring of industrial processes. |
Factors Affecting the Measurements of Conductivity Meters
By understanding these factors and applying proper calibration and maintenance practices, users can ensure more accurate and reliable conductivity measurements.
| Factor | Effect on Conductivity Measurement | Solution/Compensation |
| Temperature | Higher temperatures increase ion mobility, leading to higher conductivity readings. | Use conductivity meters with automatic temperature compensation (ATC) to correct for temperature variations. |
| Ion Concentration | Higher ion concentration results in higher conductivity; lower concentration leads to lower readings. | Select a meter with an appropriate measurement range for the expected ion concentration. |
| Electrode Condition | Dirty, corroded, or fouled electrodes can cause inaccurate readings. | Regularly clean and calibrate electrodes to maintain accuracy. |
| Solution Composition | Different ions contribute differently to conductivity (e.g., NaCl vs. CaCl₂). | Consider the specific ion composition when interpreting conductivity values. |
| Polarization Effects | Occurs when DC voltage is applied, causing inaccurate readings. | Use an alternating current (AC) source to prevent electrode polarization. |
| Interfering Substances | Organic compounds, oils, or suspended particles can affect conductivity. | Use filtration or pre-treatment if necessary before measurement. |
| Cell Constant Variations | The distance between electrodes and their surface area can change due to wear or manufacturing differences. | Regularly calibrate the meter using standard conductivity solutions. |
Summary
Conductivity meters are invaluable tools for assessing the ionic content of liquids. By measuring the electrical conductivity of a solution, these devices provide critical insights into water purity, chemical concentrations, and industrial process control. Understanding their working principles and influencing factors allows for more precise and reliable measurements.